Tooth regeneration in 2026
Regrowing teeth is no longer just a lab experiment. Most previous attempts relied on stem cells and growth factors to nudge the body into self-repair, but results were rarely consistent. We’ve struggled to find enough high-quality stem cells or get them to actually form the complex layers of a real tooth.
Gene therapy changes the strategy by giving cells direct instructions to build. By introducing specific genetic material, we can flip the switches that trigger tooth development. It is a difficult process, but it offers a permanent fix for tooth loss that dentures can't match.
The National Institute of Dental and Craniofacial Research (NIDCR) is a major funder of this research, recognizing the potential to revolutionize oral health care. They've invested heavily in projects exploring gene delivery systems and the identification of key genes involved in tooth formation. This funding is critical, as the development of gene therapies is a lengthy and expensive process, requiring years of research and rigorous testing.
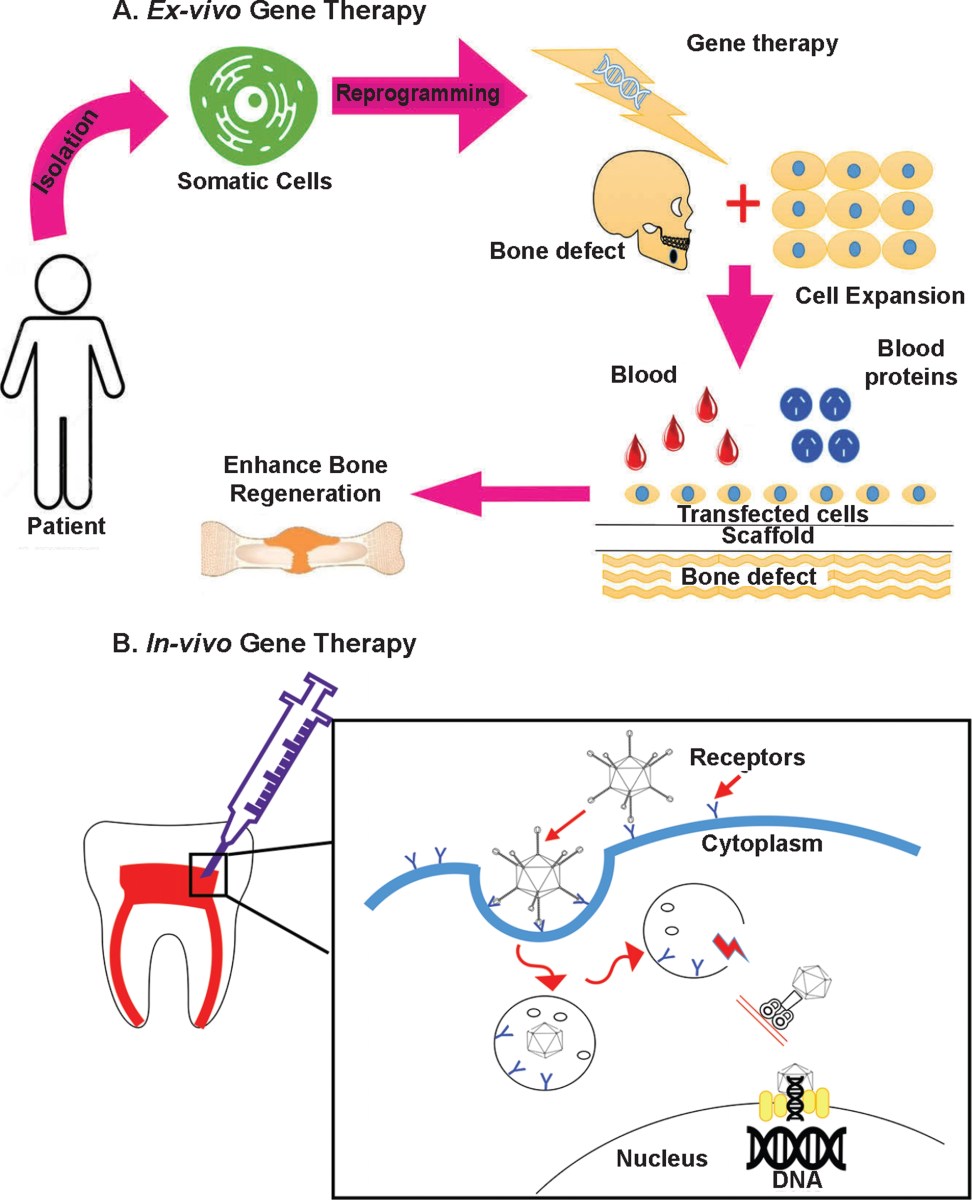
How gene therapy works
At its core, gene therapy for tooth regeneration aims to deliver specific genes to cells within the tooth bud or surrounding tissues, prompting them to rebuild lost tooth structures. The genes targeted are those that play critical roles in odontogenesis – the development of teeth. This includes genes regulating enamel formation (like amelogenin), dentinogenesis (such as DSPP – dentin sialoprotein), and periodontal ligament development (COL1A1 and COL1A2).
Delivering these genes effectively requires a vector – a carrier molecule that transports the genetic material into the target cells. Viral vectors, particularly adeno-associated viruses (AAVs), are frequently used due to their ability to infect cells efficiently and their relatively low immunogenicity. However, they aren’t without drawbacks; they can elicit an immune response and have limited cargo capacity. Nanoparticles, offering greater control over gene delivery and reduced immunogenicity, are also being explored.
Research methodology, as detailed in publications from pmc.ncbi.nlm.nih.gov, emphasizes the importance of robust in vitro and in vivo models to assess the efficacy and safety of gene therapy approaches. Careful consideration must be given to vector design, gene expression levels, and the potential for off-target effects. The goal is to achieve precise and sustained gene expression within the tooth-forming cells.
Choosing a vector is a balancing act. AAVs are reliable but have tiny 'cargo holds,' forcing us to be picky about which genes we send. Nanoparticles can carry much more, but they aren't as good at actually getting into the cells yet.
Recent breakthroughs from 2024 to 2026
The past few years have seen significant progress in gene therapy for tooth regeneration, particularly in pre-clinical animal studies. Researchers at the University of Pennsylvania, led by Dr. Songtao Shi, published findings in Nature (2025) demonstrating the successful regrowth of entire teeth in pigs using a modified AAV vector to deliver the BMP4 gene. This gene is crucial for initiating tooth development.
Another notable breakthrough came from the Institute of Craniofacial Biology at the University of Southern California. Dr. Yang Chai’s team reported in Developmental Cell (2026) that they were able to regenerate tooth root structures in dogs by delivering the Wnt1 gene using a collagen scaffold. This resulted in the formation of functional periodontal ligament attachment, crucial for tooth stability.
Researchers at Kyoto University in Japan have focused on enamel regeneration. In a study published in Scientific Reports (2024), they demonstrated the ability to enhance enamel formation in mice by delivering the Enamelin gene via nanoparticles. While the regenerated enamel wasn’t a perfect match to natural enamel in terms of hardness, it showed significantly improved resistance to acid erosion.
These studies aren’t simply showing some regeneration; they're demonstrating the potential for functional tooth structures. The pig study, for example, showed that the regrown teeth were able to withstand normal chewing forces. The dog study showed proper root attachment, and the mouse study showed improved enamel resistance. These are specific, measurable outcomes.
Current clinical trials
As of late 2026, there are no large-scale, fully approved human clinical trials for gene therapy-based tooth regeneration. Several Phase 1 safety trials are underway, primarily focused on assessing the feasibility of delivering gene therapy vectors to the periodontal tissues. One such trial, sponsored by a biotech company called DentRegen, is recruiting patients with localized periodontal defects. The inclusion criteria require patients to have at least one tooth with significant bone loss around the root.
The ADA Clinical Practice Guidelines emphasize the importance of careful patient selection and informed consent in any clinical trial involving novel therapies. DentRegen’s protocol involves a single injection of an AAV vector carrying the VEGF gene (vascular endothelial growth factor) into the affected area. VEGF is intended to promote blood vessel formation and tissue regeneration. Preliminary data presented at the International Association for Dental Research (IADR) conference in 2026 showed promising signs of bone regeneration in a small cohort of patients, but long-term efficacy remains to be determined.
The major hurdles preventing wider clinical translation include safety concerns – particularly the potential for off-target effects and immune responses – and the high cost of production. Regulatory approval is another significant challenge, as gene therapies are subject to stringent review by agencies like the FDA. The cost of manufacturing these therapies is substantial, potentially limiting access to those who can afford it.
New ways to deliver genes
While direct injections are the most common method for delivering gene therapy vectors, researchers are exploring more sophisticated approaches to improve targeted delivery and sustained gene expression. Biomaterials, such as collagen scaffolds and hydrogels, can serve as carriers for gene-carrying vectors, providing a localized and sustained release of the therapeutic genes.
3D-printed structures are also gaining traction. These structures can be customized to fit the specific defect site and provide a framework for tissue regeneration. Researchers are experimenting with incorporating gene-delivering vectors directly into the 3D-printed material, allowing for precise control over gene delivery. This is particularly useful when attempting to regenerate complex tooth structures.
The advantages of these alternative delivery methods include improved targeting, reduced off-target effects, and prolonged gene expression. However, challenges remain, such as ensuring biocompatibility of the materials and achieving sufficient gene transfer efficiency. Precise control over the degradation rate of the biomaterial is also crucial to ensure optimal tissue regeneration.
Safety and ethics
Gene therapy is not without potential risks. Off-target effects – where the gene therapy vector delivers the therapeutic gene to unintended cells – are a major concern. This could lead to unwanted gene expression and potentially harmful consequences. Immune responses to the viral vector or the newly expressed protein can also occur, potentially triggering inflammation or even organ damage.
Insertional mutagenesis, while rare, is another potential risk associated with viral vectors. If the vector integrates into the host genome in a critical location, it could disrupt normal gene function and potentially lead to cancer. Stringent safety testing and careful vector design are essential to minimize these risks. Regulatory oversight is also crucial.
Germline gene therapy – altering the genes in reproductive cells – raises significant ethical concerns. While not currently being pursued for tooth regeneration, the potential for heritable changes necessitates careful consideration. Long-term monitoring of patients who receive gene therapy is essential to detect any delayed adverse effects. Current regulations prioritize somatic gene therapy – targeting non-reproductive cells – to avoid these ethical dilemmas.
Who will pay for it?
The cost of gene therapy for tooth regeneration is likely to be substantial, at least initially. The manufacturing process is complex and expensive, requiring specialized facilities and expertise. The cost will likely be driven by factors such as vector production, quality control, and personalized treatment protocols.
It’s unlikely that gene therapy for tooth regeneration will be readily accessible to the average patient in the near future. Initially, it will likely be a niche treatment reserved for those with significant resources or for patients with rare genetic conditions causing severe tooth defects. The role of insurance coverage and government funding will be critical in determining broader access.
The economic factors influencing the cost include the scale of production, competition among manufacturers, and the development of more efficient manufacturing processes. As the technology matures and production costs decrease, the price may become more affordable, potentially broadening access to this potentially transformative therapy.



No comments yet. Be the first to share your thoughts!