The shift from restoration to regeneration
For decades, the field of dentistry has focused on restoring damaged teeth – fillings, crowns, implants, and bridges have been the standard of care. But what if we could actually regenerate lost tooth structure? That’s the promise driving a significant shift in dental research toward gene therapy. We’re seeing a move away from simply replacing teeth to biologically rebuilding them.
Traditional restorative methods, while effective, aren’t without limitations. Implants require surgery and can be costly, while bridges impact adjacent teeth. Fillings, even advanced composite ones, are susceptible to failure and require eventual replacement. The goal of tooth regeneration is to offer a permanent, biologically sound solution, mirroring the natural healing processes of the body.
Complete tooth regeneration is still a massive hurdle, but gene therapy is moving from theory to actual practice. We are seeing the first real pathways to growing biological tooth structure rather than just drilling and filling.
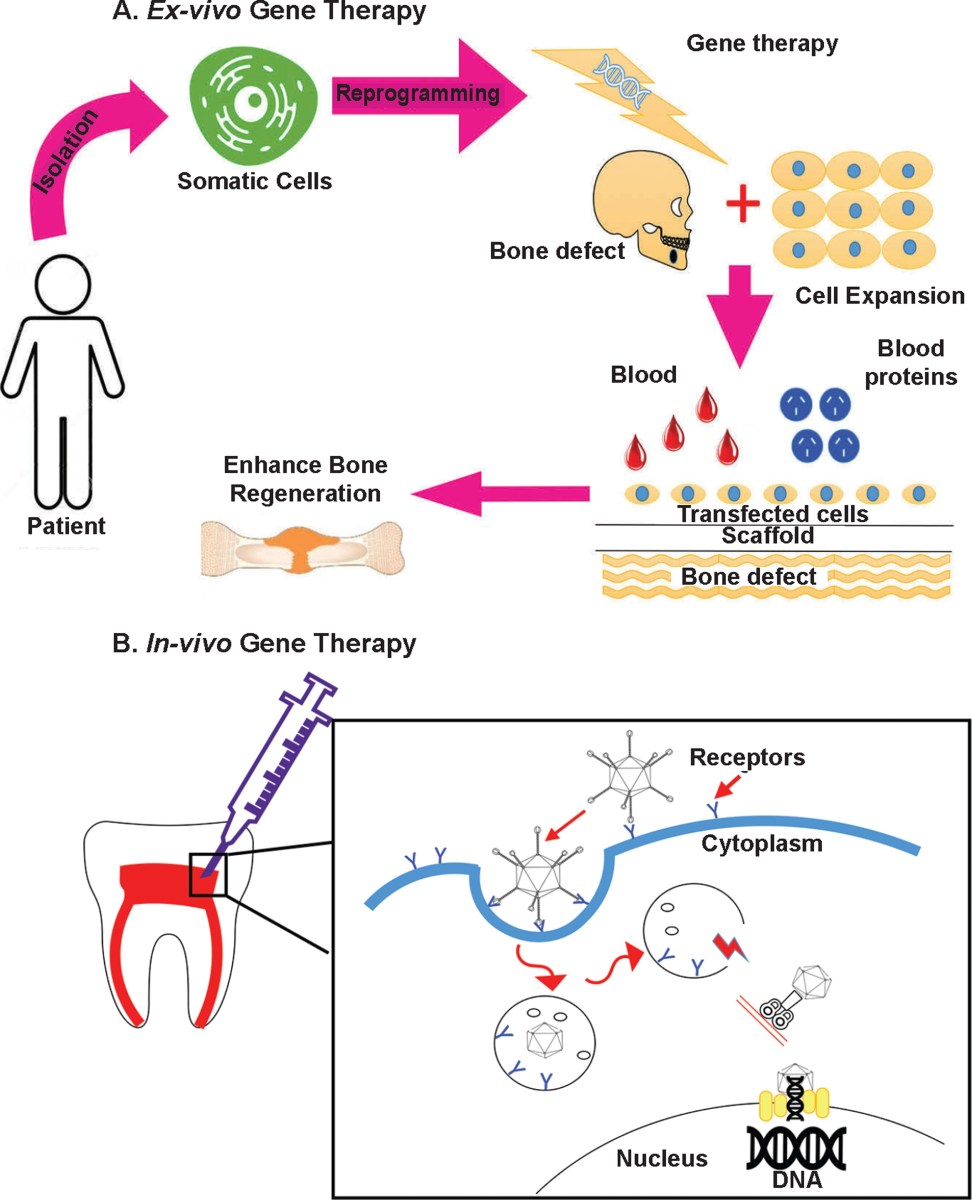
The genes and vectors building new teeth
The foundation of gene therapy for tooth regeneration lies in understanding the genes that orchestrate tooth development. Several genes have emerged as key players. Msx1, for example, is crucial for the initiation of tooth formation. Pax9 is essential for the development of the dental mesenchyme, the tissue that gives rise to the tooth’s supporting structures. And Runx2 plays a vital role in the differentiation of odontoblasts, the cells that produce dentin.
Manipulating the expression of these genes – essentially turning them on or off at specific times – can stimulate the formation of new dentin and enamel. But getting these genes into the right cells is the challenge. This is where vectors come in. Viral vectors, like adeno-associated viruses (AAVs) and lentiviruses, are highly efficient at delivering genetic material.
AAVs are favored for their low immunogenicity and ability to infect a wide range of cell types, but they have a limited cargo capacity. Lentiviruses can carry larger genes, but they have a higher risk of insertional mutagenesis. Non-viral vectors, such as nanoparticles, offer a safer alternative, but their efficiency is generally lower. Researchers are constantly working to improve vector design and targeting to maximize efficacy and minimize side effects.
The choice of vector is a careful balancing act. It depends on the specific gene being delivered, the target cells, and the desired duration of gene expression. There isn’t a one-size-fits-all solution; each application requires a tailored approach.
What we've learned from animal models
Significant progress has been made in preclinical studies using animal models. Researchers at the National Institute of Dental and Craniofacial Research (NIDCR) have demonstrated successful dentin regeneration in mice using AAV-mediated delivery of Runx2. Published in the Journal of Dental Research (2024), the study showed substantial dentin formation within damaged root canals.
A team at the University of Pennsylvania, led by Dr. Songtao Shi, reported promising results in pigs using a combination of gene therapy and a biodegradable scaffold. They were able to regenerate a significant portion of tooth structure, including dentin and enamel, after inducing tooth damage. The research, published in Scientific Reports (2025), highlighted the importance of providing a supportive microenvironment for regeneration.
Success in mice doesn't always mean success in humans. Larger mammals like pigs and dogs have more complex tooth structures and different immune responses that can block regeneration. Dog studies are the most reliable indicator for human results, though they take much longer to complete.
Recent work at King’s College London (PMC, 2026) focused on using gene-activated stem cells to regenerate periodontal tissues in dogs. This demonstrated the potential for gene therapy to address not just tooth structure, but also the supporting tissues essential for tooth stability. The research showed significant regeneration of cementum, periodontal ligament and alveolar bone.
- NIDCR (2024): Runx2 gene therapy in mice for dentin regeneration.
- University of Pennsylvania (2025): Combined gene therapy & scaffold for tooth structure regeneration in pigs.
- King’s College London (2026): Gene-activated stem cells for periodontal tissue regeneration in dogs.
Preclinical Studies in Gene Therapy for Tooth Regeneration (as of late 2023/early 2024)
| Animal Model | Gene(s) Used | Vector Type | Regeneration Level | Key Findings |
|---|---|---|---|---|
| Mouse | BMP2, Runx2 | Adeno-associated virus (AAV) | Partial | Demonstrated localized dentin formation in pulp cavities; limited root structure development. |
| Rat | Dlx1, Dlx2, Msx1 | Retroviral vector | Partial | Induced formation of dental papilla-like structures, but complete tooth formation was not achieved. |
| Pig | Wnt3a, Shh | Lentiviral vector | Complete cusp | Successful regeneration of cusp-like structures on extracted teeth, showing potential for crown formation. |
| Mouse | Amelogenin | Plasmid DNA (electroporation) | Partial | Enhanced dentin bridge formation in exposed pulp, suggesting potential for pulp protection and repair. |
| Rabbit | BMP4, FGF | Adenoviral vector | Partial | Promoted cementum and periodontal ligament regeneration in root resorption models. |
| Dog | BMP2, TGF-β1 | AAV | Partial | Observed alveolar bone regeneration alongside limited tooth structure development following tooth extraction. |
| Mouse | Scleraxis | Adeno-associated virus (AAV) | Partial | Showed enhanced dental mesenchymal stem cell differentiation and increased expression of dental markers. |
Illustrative comparison based on the article research brief. Verify current pricing, limits, and product details in the official docs before relying on it.
Current clinical trials
As of late 2026, clinical trials for tooth regeneration using gene therapy are still in the early stages. Several Phase 1 trials are underway, primarily focused on assessing the safety and feasibility of delivering genes to damaged teeth. These trials typically involve a small number of patients with localized tooth damage, such as root canal defects or small enamel lesions.
Inclusion criteria generally focus on patients with healthy overall health and no pre-existing autoimmune conditions. Exclusion criteria often include pregnancy, smoking, and the use of immunosuppressant medications. The primary endpoints being measured are safety – monitoring for adverse events – and the ability to detect gene expression in the target tissues.
Moving these therapies into widespread clinical use faces several hurdles. Safety remains the top concern. Ensuring that the genes are delivered specifically to the target cells and don’t cause off-target effects is critical. Efficacy is another challenge. Achieving consistent and predictable regeneration is essential. Finally, cost is a significant factor. Gene therapy is currently expensive, and making it accessible to a wider population will require significant cost reductions.
I anticipate that Phase 2 trials, evaluating efficacy in a larger patient population, will begin to emerge in 2027 or 2028. These trials will be pivotal in determining whether gene therapy can truly deliver on its promise of tooth regeneration.
Beyond Whole Tooth: Targeted Regeneration
The potential of gene therapy extends beyond regenerating entire teeth. Researchers are exploring its use in targeted regeneration – repairing specific damaged tissues within the mouth. One promising area is enamel repair. Enamel, unlike other tissues, lacks the ability to regenerate. Gene therapy could potentially stimulate the formation of new enamel crystals, addressing cavities and preventing tooth decay.
Another application is in the treatment of periodontal disease. By delivering genes that promote the regeneration of cementum, periodontal ligament, and alveolar bone, it may be possible to rebuild the supporting structures of the teeth, reversing the effects of gum disease. This could offer a less invasive alternative to traditional periodontal surgery.
Furthermore, gene therapy can be used to enhance the success of traditional dental procedures. For example, researchers are developing bio-root implants coated with genes that stimulate tissue integration, improving the long-term stability of the implant. This represents a powerful synergy between conventional dentistry and cutting-edge genetic technologies.
Safety and ethics
Gene therapy, like any medical intervention, carries potential risks. Off-target effects – the delivery of genes to unintended cells – are a major concern. Immune responses to the viral vectors can also occur, leading to inflammation and tissue damage. The risk of insertional mutagenesis – the insertion of the gene into a location in the genome that disrupts normal gene function – is another potential complication.
Careful vector design and targeted delivery strategies are crucial to minimizing these risks. Rigorous safety testing in preclinical models is also essential before moving to clinical trials. Long-term monitoring of patients who undergo gene therapy is needed to assess the potential for delayed adverse effects.
Germline therapy—changing genes passed to children—is a different beast. Dentistry isn't touching that yet, but the tech raises questions about where we draw the line. People are naturally nervous about genetic modification, so the science needs to be transparent rather than hidden behind jargon.



No comments yet. Be the first to share your thoughts!