Regenerating tissue instead of patching it
For decades, dentistry has largely focused on restoring oral health – patching up damage with fillings, replacing missing teeth with implants, and managing disease. But what if we could go further? Regenerative dentistry represents a fundamental shift in how we approach oral health care, moving beyond repair to actually rebuilding tissues. This isn't science fiction anymore; it’s an evolving field with the potential to revolutionize patient care.
Traditional treatments, while effective, have limitations. Fillings can fail, requiring replacement, and implants, though successful in many cases, don’t fully replicate the natural structure and function of a tooth. They also require a degree of bone support that isn't always present. Regenerative dentistry aims to address these shortcomings by harnessing the body’s own healing mechanisms to restore lost or damaged tissues.
The core idea is to stimulate the body to regenerate what’s been lost – bone, enamel, dentin, and even the periodontal ligament. This involves a complex interplay of biomaterials, growth factors, stem cells, and, increasingly, gene therapy. It’s a move towards treatments that are more natural, more durable, and ultimately, better for the patient.
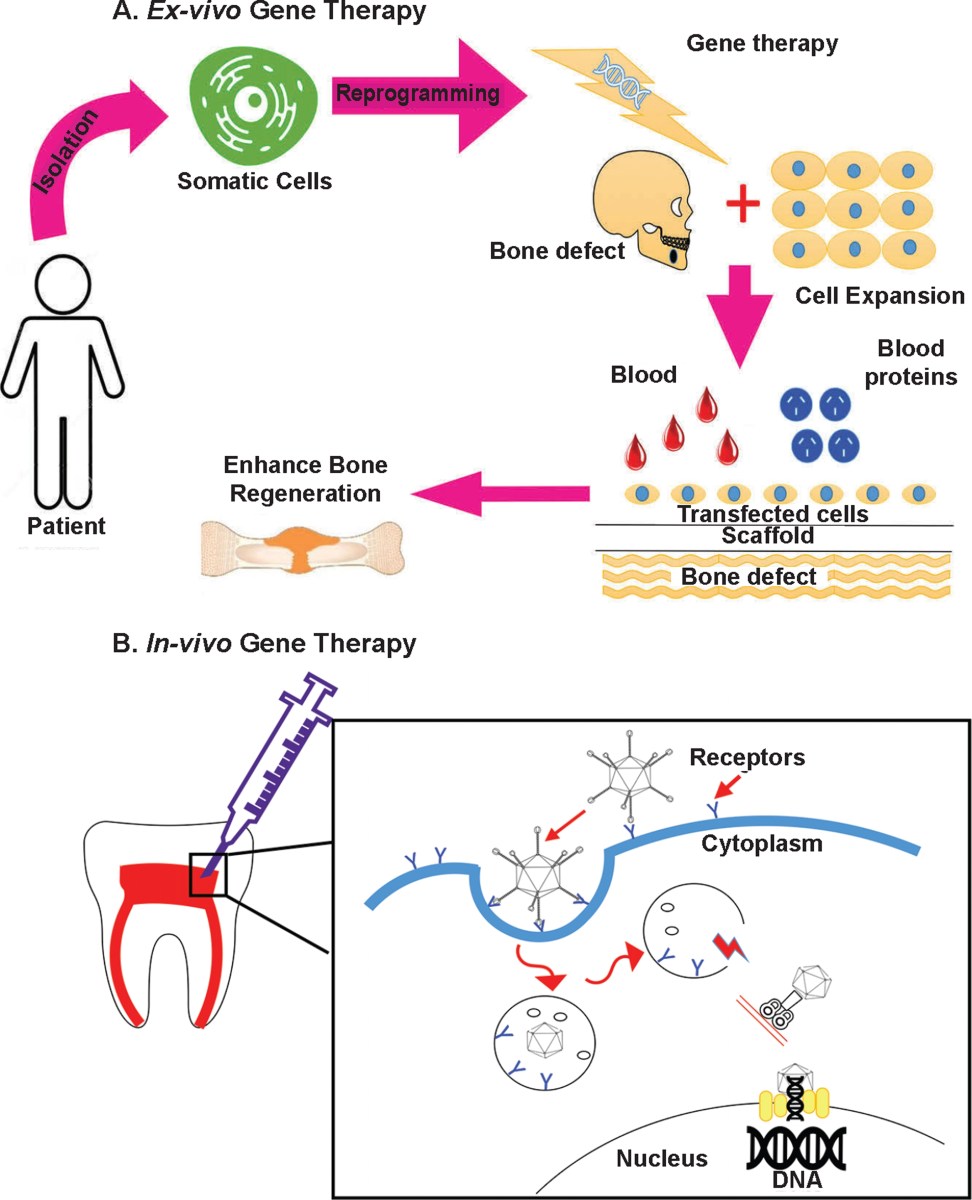
Using genes to grow teeth
One of the most exciting areas within regenerative dentistry is the potential to actually grow back teeth. This isn’t about simply repairing damage, but about reactivating the developmental pathways that created teeth in the first place. Researchers are focusing on genes critical to tooth formation, like ameloblastin and enamelin, which are responsible for enamel and dentin development, respectively.
The approach involves delivering these genes directly to cells within the tooth bud or surrounding tissues. Currently, the most promising tools for this delivery are viral vectors – modified viruses that can carry genetic material into cells – and CRISPR-Cas9 gene editing technology. CRISPR allows for precise targeting and modification of genes, offering the possibility of correcting genetic defects that contribute to tooth loss or malformation.
Animal trials show promise, but I am not sure when we will see these results in humans. The NIDCR is currently testing how to control the growth so we don't end up with disorganized tissue. The main problem is still getting the genes into the right cells safely.
Bioactive Materials and Scaffolds
Gene therapy and stem cell therapies often require a supportive environment for cells to grow and organize into functional tissues. This is where bioactive materials and scaffolds come into play. These materials act as a three-dimensional framework, guiding tissue regeneration and providing structural support.
A variety of materials are being investigated, including collagen, hydrogels, ceramics like calcium phosphate, and even synthetic polymers. Collagen, a natural protein, is biocompatible and promotes cell adhesion. Hydrogels, with their high water content, mimic the natural extracellular matrix, providing a favorable environment for cell proliferation. Ceramics are often used for bone regeneration due to their osteoconductive properties.
These scaffolds are active. We can coat them in growth factors to tell cells what to do. The material has to be something the body won't reject, and it needs to dissolve once the new tissue is strong enough to stand on its own.
Scaffold Material Comparison for Regenerative Dentistry Applications
| Material Type | Biocompatibility | Biodegradability | Mechanical Strength | Manufacturing Complexity |
|---|---|---|---|---|
| Collagen | Excellent – naturally occurring in the body | High – degrades relatively quickly | Moderate – can be improved with crosslinking | Moderate – sourcing and purification can be involved |
| Hydrogels | Good to Excellent – tunable for specific cell interactions | Tunable – degradation rate can be controlled | Low to Moderate – generally weaker than other options | Moderate – polymerization process requires careful control |
| Ceramics (e.g., Hydroxyapatite) | Good – osteoconductive properties promote bone integration | Slow – degradation occurs over a longer period | High – provides structural support | High – requires high-temperature processing |
| Alginate | Good – derived from natural sources | Fast – degrades quickly, limiting long-term support | Low – brittle and lacks significant strength | Low – simple gelation process |
| Chitosan | Good – antibacterial properties, promotes healing | Moderate – degradation rate is pH dependent | Moderate – can be enhanced with blending | Moderate – requires deacetylation process |
| Polycaprolactone (PCL) | Good – generally well-tolerated | Slow – very slow degradation, suitable for long-term applications | Moderate to High – can be tailored based on molecular weight | Moderate – requires solvent casting or melt processing |
Qualitative comparison based on the article research brief. Confirm current product details in the official docs before making implementation choices.
Stem Cell Therapies in Oral Tissues
Stem cells possess the remarkable ability to differentiate into various cell types, making them a powerful tool in regenerative dentistry. Several types of stem cells are being explored for oral tissue regeneration, including dental pulp stem cells (DPSCs) found within teeth, periodontal ligament stem cells (PDLSCs) which support tooth attachment, and bone marrow stem cells. DPSCs and PDLSCs are particularly attractive because they are readily accessible from the oral environment.
The process typically involves harvesting these stem cells, expanding their numbers in a laboratory setting, and then differentiating them into specific cell types needed for regeneration – such as odontoblasts (which form dentin) or cementoblasts (which form cementum). These differentiated cells can then be implanted into the damaged area to promote tissue repair. For example, PDLSCs have shown promise in regenerating periodontal tissues lost due to gum disease.
However, stem cell therapies aren’t without challenges. Immune rejection is a concern, particularly when using allogeneic stem cells (from a donor). Long-term stability of the regenerated tissue is another consideration. Ensuring that the new tissue integrates properly with the surrounding structures and remains functional over time requires careful control of the differentiation process and a supportive microenvironment. Clinical trials are underway to address these issues.
One promising avenue is using the patient’s own stem cells – autologous transplantation – to minimize the risk of rejection. Researchers are also investigating ways to modify stem cells to enhance their regenerative potential and improve their survival in the host tissue.
Saliva tests and custom treatment
Regenerative dentistry isn’t a one-size-fits-all approach. Individual patient factors – genetics, overall health, lifestyle – all play a role in how they respond to treatment. This is where salivary diagnostics comes in. Analyzing saliva provides a non-invasive way to assess a patient’s oral health status and predict their response to regenerative therapies.
Saliva contains a wealth of biomarkers – proteins, enzymes, DNA, and RNA – that can provide insights into a patient’s genetic predisposition to dental diseases, their inflammatory response, and their overall immune function. This information can be used to tailor regenerative therapies to the individual patient, maximizing their chances of success. The National Institutes of Health (NIH) is investing heavily in research to develop and validate salivary diagnostic tests.
For example, identifying genetic markers associated with periodontal disease could help clinicians select the most appropriate regenerative approach for a patient with gum disease. Analyzing salivary enzymes could reveal a patient’s ability to remineralize enamel, informing the use of bioactive materials that promote enamel regeneration. This move toward personalized care promises to significantly improve the outcomes of regenerative dental treatments.
Clinical Trials and Regulatory Hurdles
Despite the tremendous progress in regenerative dentistry, these therapies are still largely in the experimental stage. Numerous clinical trials are currently underway, evaluating the safety and efficacy of various regenerative approaches for treating periodontal disease, bone loss, tooth avulsion, and enamel defects. These trials are crucial for gathering the rigorous scientific evidence needed to support widespread adoption.
However, bringing these therapies to market is not without its challenges. Regulatory approval from the Food and Drug Administration (FDA) is a significant hurdle. The FDA requires extensive data demonstrating the safety and effectiveness of new therapies before they can be approved for clinical use. The complexity of regenerative therapies – often involving cells, genes, and biomaterials – adds to the regulatory burden.
Cost-effectiveness is another critical factor. Regenerative therapies are often more expensive than traditional treatments, at least initially. Demonstrating that these therapies provide long-term value and reduce healthcare costs in the long run will be essential for gaining acceptance from insurers and patients. I think it’s realistic to anticipate a gradual rollout of these therapies, starting with niche applications where they offer a clear advantage over existing treatments.
The Economic Impact of Regenerative Dentistry
The potential economic impact of regenerative dentistry on the dental industry is substantial. While the initial cost of these therapies may be higher than traditional treatments like fillings or implants, the long-term economic benefits could be significant. By regenerating damaged tissues, we can potentially prevent tooth loss and the need for costly replacements.
The American Dental Association (ADA) is actively tracking the economic trends in regenerative dentistry. They estimate that the market for regenerative dental products and therapies could reach several billion dollars within the next decade. This growth will be driven by increasing demand for more natural and durable dental treatments, as well as advancements in regenerative technologies.
Reducing the prevalence of chronic dental diseases, such as periodontitis, could also lead to significant healthcare savings. Periodontal disease is linked to systemic health problems, such as heart disease and diabetes, and its treatment is often expensive. Regenerative therapies that effectively address the underlying causes of periodontal disease could not only improve oral health but also reduce overall healthcare costs.



No comments yet. Be the first to share your thoughts!